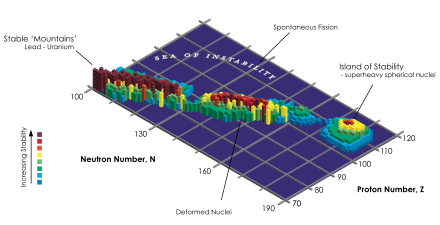
I'll take a crack at it. Big takeaway: you know those two rows on the bottom of the period table that are separated from the rest of the elements? Those are lanthanide and actinide elements. They are massive - like really massive (for an atom). Being big is cool, but it also makes you very unstable. In this case, unstable means you aren't long for this world. Once created (and most of them are only created because we've forced them into existence, meaning they don't occur naturally), they only live for minutes or less. Sometimes just fractions of a second. Some of these elements have properties that would be SUPER interesting, but because they only happen artificially and they don't last long, there isn't much use in them.
Fast forward to theory time. The 'island of stability' is this mythical level of big atoms beyond even those we've created. They would be stable long enough to be studied and useful. To be clear, we've never actually seen any of these atoms and it's questioned if they did exist, would they be stable. But the hope is that one day we'll find them or be able to create them and usher in some wicked new science.
Fun fact: finding any of these big boys out in the cosmos is a
great way to search for aliens, as we know of no methods by which they can be produced naturally.
Island of stability chart. I like it because it looks like Hawaii. Keep in mind we haven't seen this island yet. It's that last little section of red on the far right - red meaning stable.
View attachment 3955
Fun video from Dr. Kipping (one of my favorite dudes in astronomy).

 charmingscience.com
charmingscience.com

 charmingscience.com
charmingscience.com